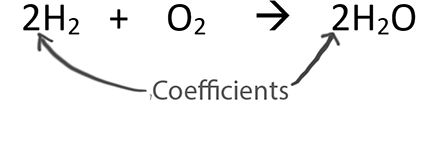3g of H2 reacts with 29 g of O2 to give H2O.Find i) Limiting reagent ii)Max amount of H2O formed iii) Amount of reactants which remains unreacted.

n mole each of h2o h2 o2 r tken in closed container at temperature t if y mole of h2 r disasssociated at equillibrium n equillibrium pressure is p the cgt66gee -Chemistry -
Kp for the reaction CO2 + H2 =CO + H2O is found to be 16 at a given temperature. Originally equal number of moles of H2 and CO2 were placed in the

H2 Evolution from H2O via O–H Oxidative Addition Across a 9,10-Diboraanthracene | Inorganic Chemistry | ChemRxiv | Cambridge Open Engage








![PDF] Initiation in H2/O2: Rate constants for H2+O2→H+HO2 at high temperature | Semantic Scholar PDF] Initiation in H2/O2: Rate constants for H2+O2→H+HO2 at high temperature | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/59651c1a79d5be73d45e3b492f6f4396965dd05f/5-Table1-1.png)