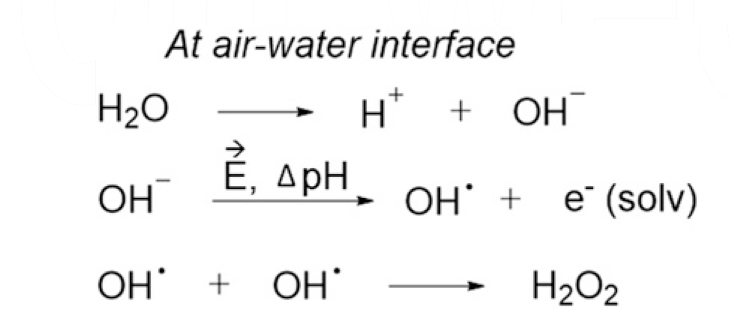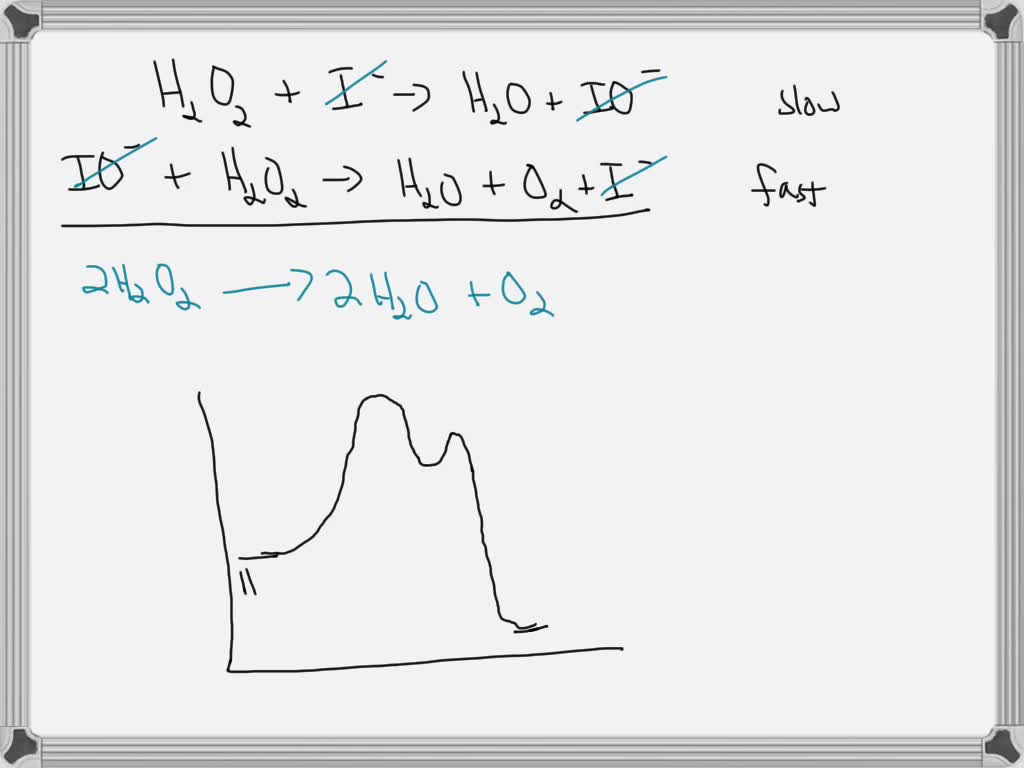
SOLVED: Step 1: H2O2 + I- → H2O + IO- (slow) Step 2: H2O2 + IO- → H2O + O2 + I- (fast) a.) Identify the rate determining step. b.) Draw a
Why is the answer B? Can someone explain this to me and why other options are incorrect. I assumed that H2O2 will decompose rapidly to form H20 and O2 with MnO2 as
1) H2O2 + O3 → H2O +2O2 2)H2O2 +Ag2O →2Ag +H2O +O2 Determine whether H2O2 is oxidised or reduced in the above reaction? Explain.

The decomposition of H2O2 has a strong thermodynamic driving force 2H2O2→ 2H2O + O2(g) Δ H = - 99kj/mole,Δ s = + 69JK^-1 mole^-1 Addition of solution of KI causes H2O2 to
![Generation of HO• Radical from Hydrogen Peroxide Catalyzed by Aqua Complexes of the Group III Metals [M(H2O)n]3+ (M = Ga, In, Sc, Y, or La): A Theoretical Study | ACS Catalysis Generation of HO• Radical from Hydrogen Peroxide Catalyzed by Aqua Complexes of the Group III Metals [M(H2O)n]3+ (M = Ga, In, Sc, Y, or La): A Theoretical Study | ACS Catalysis](https://pubs.acs.org/cms/10.1021/cs400155q/asset/images/medium/cs-2013-00155q_0021.gif)
Generation of HO• Radical from Hydrogen Peroxide Catalyzed by Aqua Complexes of the Group III Metals [M(H2O)n]3+ (M = Ga, In, Sc, Y, or La): A Theoretical Study | ACS Catalysis

H2O2=H2O+O2 balance the chemical equation @mydocumentary838. h2o2=h2o+o2 balance the equation. - YouTube

SOLVED: Balance the following equation: H2O2 HzO + 02 H202 HzO + 02 2 H2O2 2 HzO + 02 2 HzO2 2 H2O + 302 H2Oz 2 HzO + 02

Kinetic studies on the reaction of 2 with H2O2 in buffered MeCN/H2O... | Download Scientific Diagram
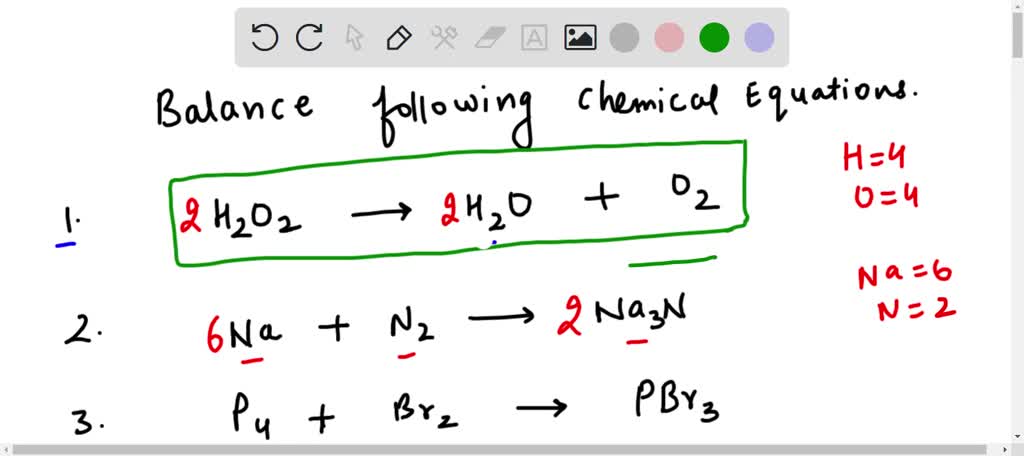
SOLVED: Balance the following chemical equations. H2O2 à H2O + O2 Na + N2 à Na3N P4 + Br2 à PBr3 O2 + Na à Na2O