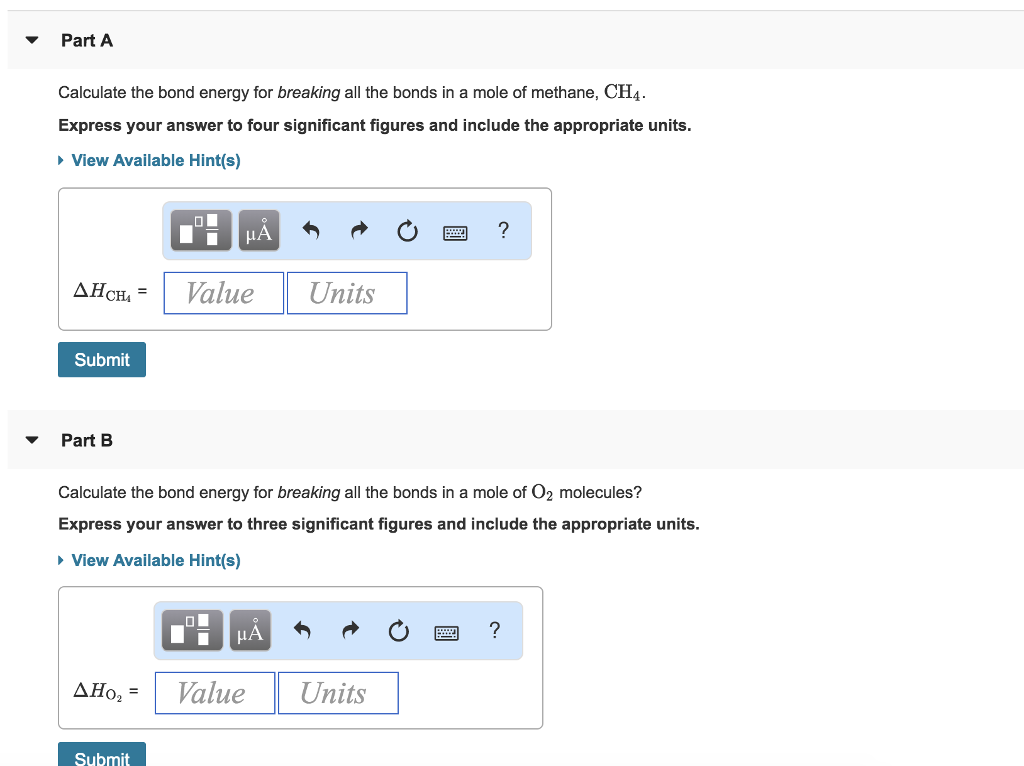
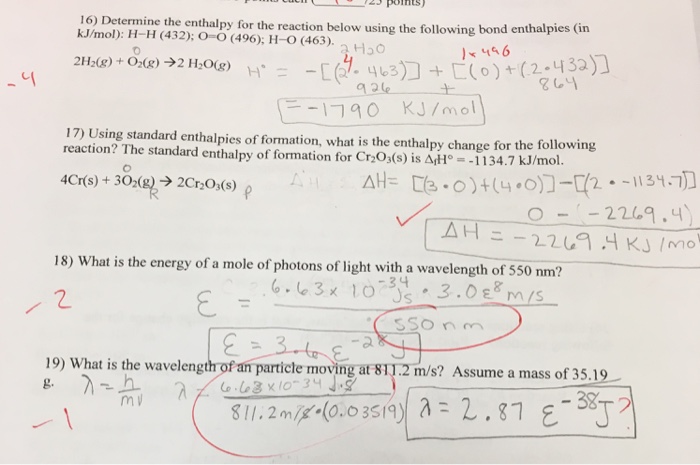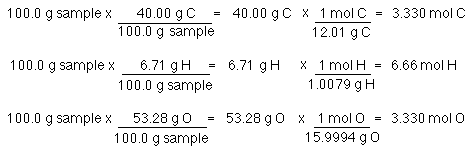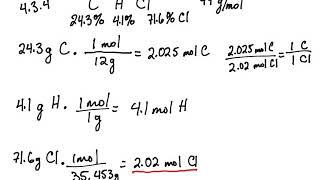SMC: Chem 11: Week 4, Chapter 3 Combustion Analysis: 91*, 92*, 94*, 138* -Scholefield Flashcards | Quizlet

Mol ratio: coefficients of a balanced equation 2 H 2 + O 2 → 2 H 2 O 2 mol H 2 for every 1 mol O 2 In chemical calculations, mol ratios convert moles of. - ppt download

Question Video: Finding the Number of Molecules in a Hydrate from the Mass of the Hydrate and Water | Nagwa

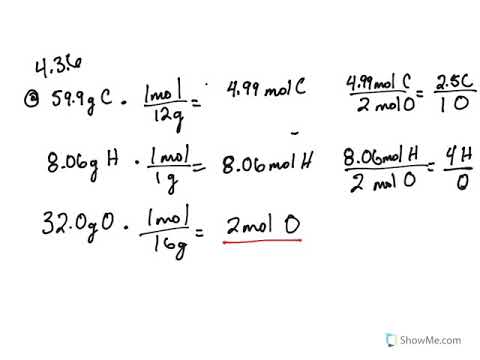
Empirical and molecular formulas for compounds that contain only carbon and hydrogen (C a H b ) or carbon, hydrogen, and oxygen (C a H b O c ) can be determined with a process called combustion analysis. The steps for this procedure are