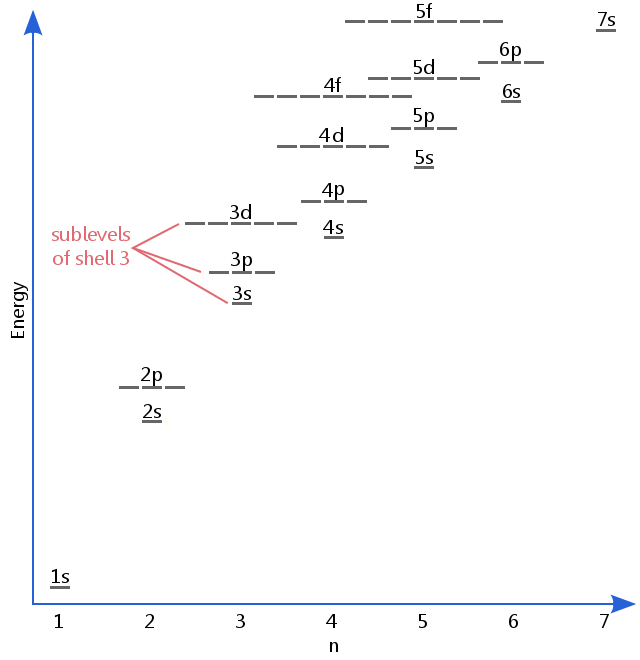
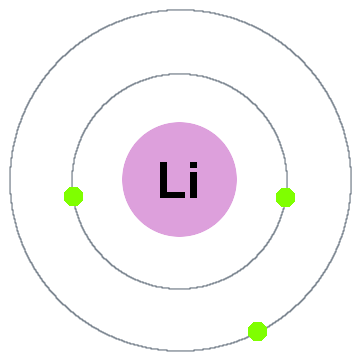
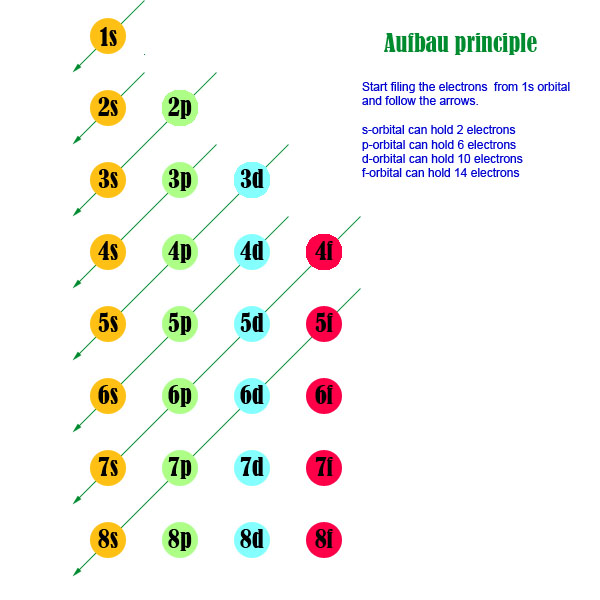
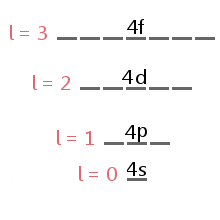Orbitals, Atomic Energy Levels, & Sublevels Explained - Basic Introduction to Quantum Numbers - YouTube
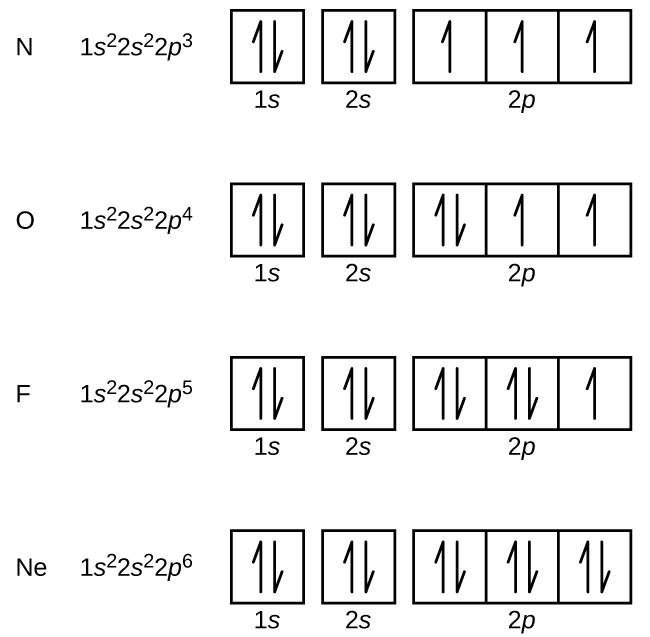
When electrons ocoupy different orbitals of the same sub-level, do they all have the same spin? | Socratic
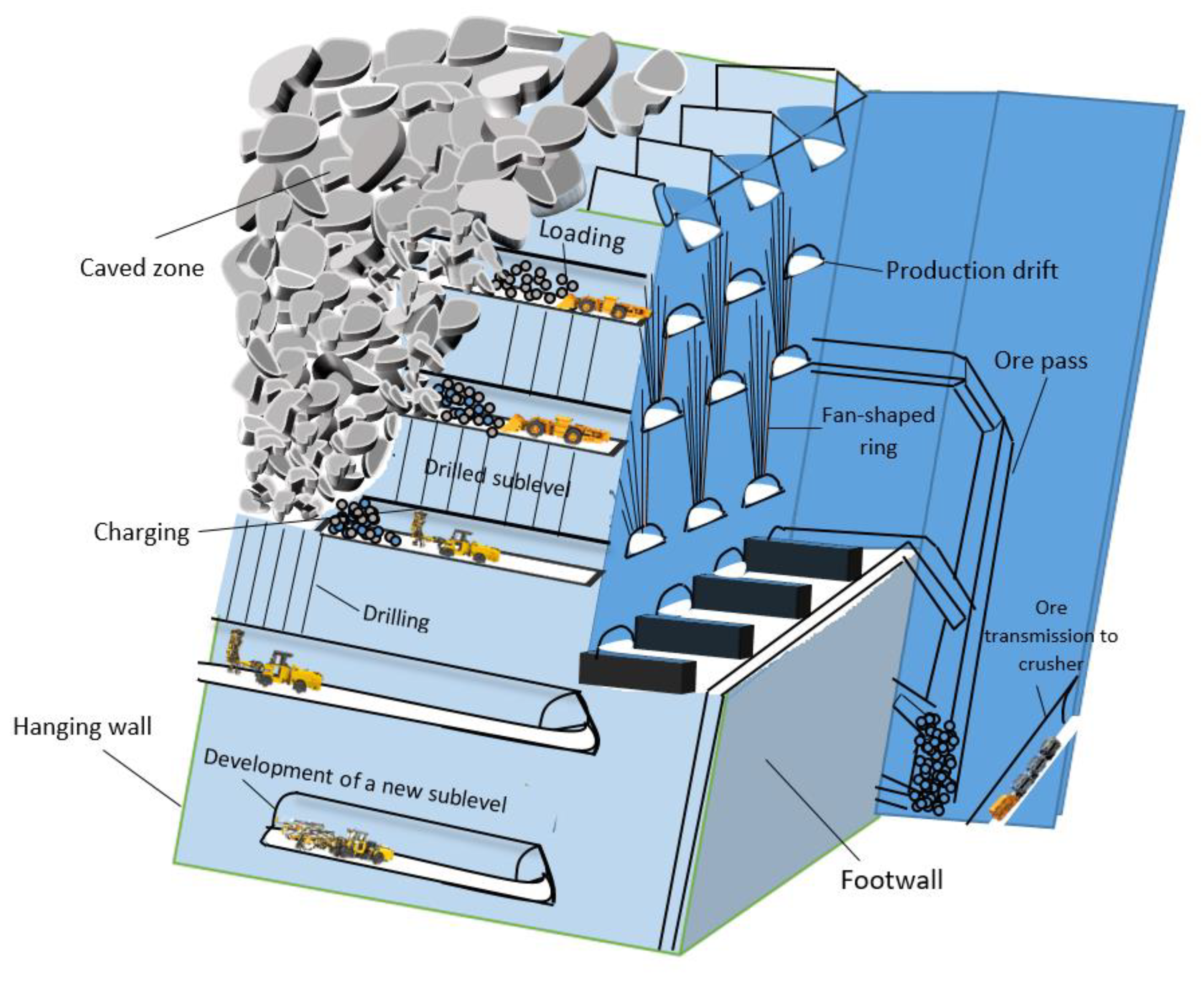
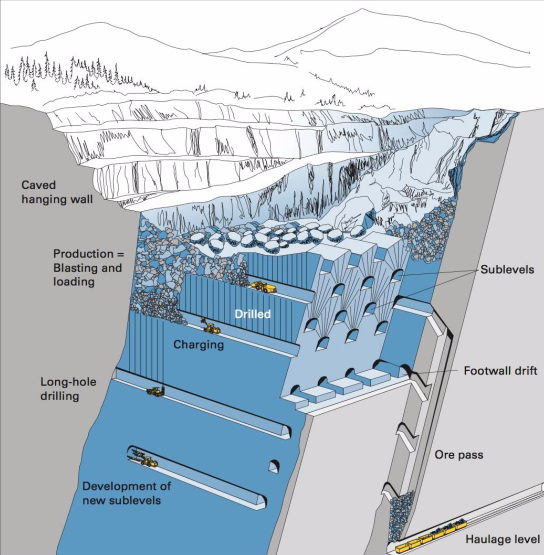
Mining | Free Full-Text | Mathematical Programming Application in Sublevel Caving Production Scheduling
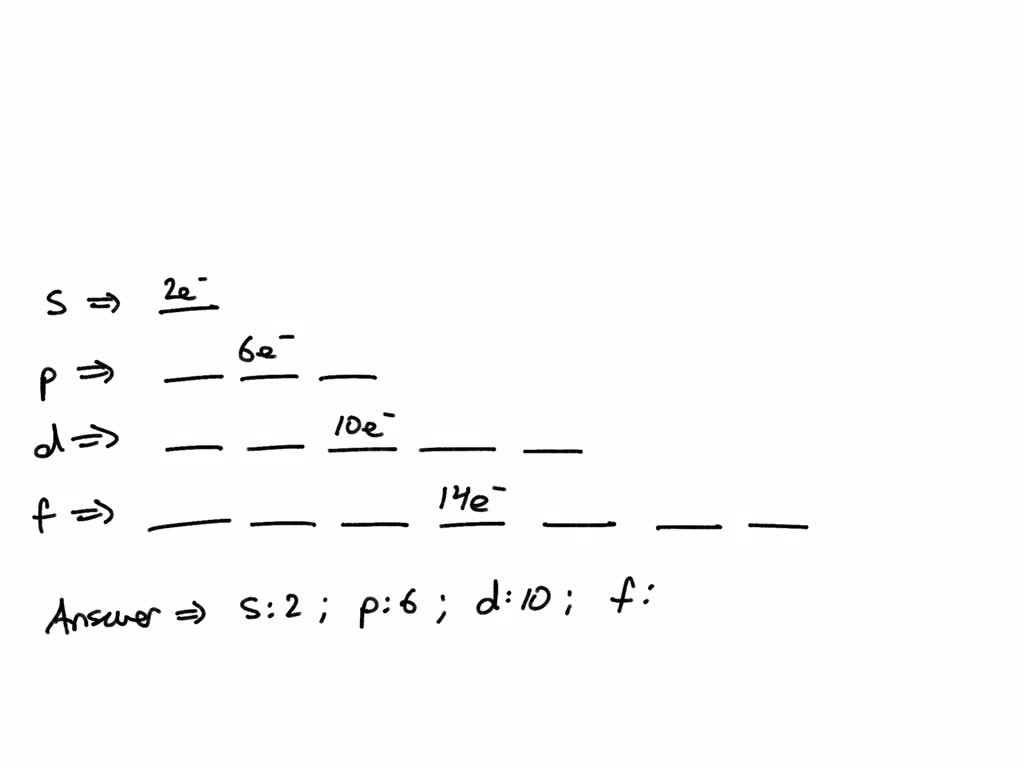
SOLVED: The letter designations for the first four sublevels with the maximum number of electrons that can be accommodated in each sublevel are Select one: a. s:2, p:4, d:6, and f:8. b.
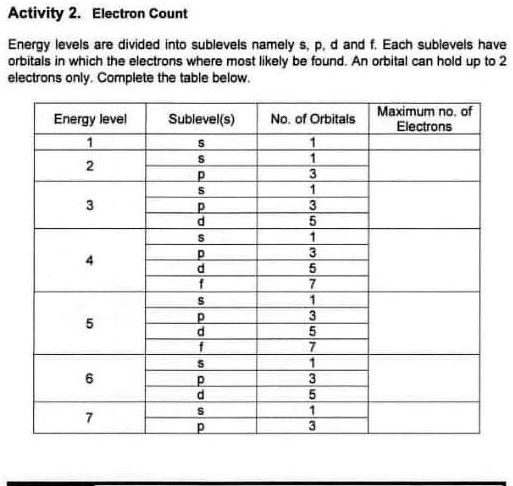
![Year 11 Chemistry] Electron Sublevels : r/HomeworkHelp Year 11 Chemistry] Electron Sublevels : r/HomeworkHelp](https://preview.redd.it/l9mlg2mhyv741.jpg?width=923&format=pjpg&auto=webp&s=930ce36a4daef652ad97cecb93f541d70c9caba7)